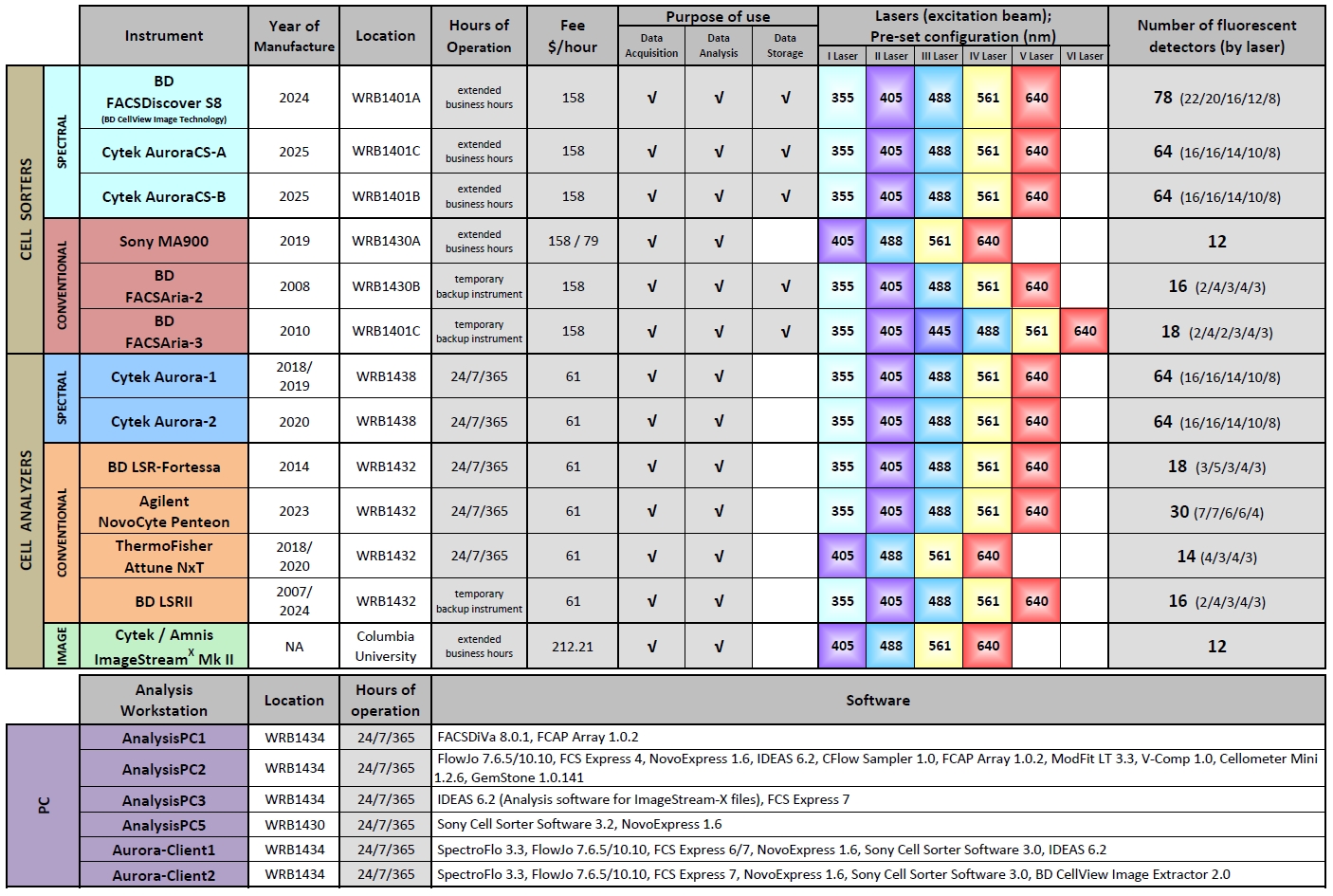
Instruments
Cytek/Amnis ImageStreamX MkII (at Columbia University)
For cell sorting, the FCRC at RU is equipped with three spectral cell sorters, one BD FACSDiscover S8 and two Cytek Aurora CS; plus one conventional cell sorter Sony MA900. The BD FACSDiscover S8, as the spectral flow cytometer sorter with sort-capable image analysis, has five lasers (355, 405, 488, 561 and 640 nm excitation wavelengths) and 78 fluorescence channels that can measure the entire emission spectra of the fluorescent dyes and sort cells based on not only fluorochrome staining but also on cell imaging features. The Cytek Aurora CS is an advanced and adaptable cell sorting system building with the power of Cytek’s Innovative Full Spectrum Profiling™ (FSP™) Technology that shares the same optical system as the Cytek Aurora analyzer with five lasers (355, 405, 488, 561 and 640 nm excitation wavelengths) and 64 fluorescence channels and can unmix up to 45 colors plus autofluorescence. The Sony MA900 is the conventional cell sorter with four lasers (405, 488, 561 and 640 nm excitation wavelengths) and 12 fluorescence channels detection. All the cell sorters are able to perform high-speed (up to 20,000 events/sec), high-purity (up to 99.9%) sterile sorts into different types of devices (tubes, plates, slides, or custom-made devices), and the spectral cell sorters can distinguish up to 45 colors in one sample and can perform six-way high-purity sort of rare cells from a high dimensional panel.
BD FACSDiscover S8 and Cytek Aurora CS sorters are operated by FCRC staff during the extended business hours. For exact time, please refer to the FCRC Cell Sorting and Instrument Training Schedule. Sony MA900 can be operated by either FCRC staff (Assisted), or researchers after completion of the required training (Unassisted).
For cell analysis purposes, the FCRC at RU is equipped with five benchtop analyzers, which cover the wide range of researcher’s needs:
The two spectral analyzers Cytek Aurora have five lasers (355, 405, 488, 561 and 640 nm) and 64 fluorescence channels. It has a unique capability of measuring the entire emission spectra of the fluorescent dyes excited by multiple lasers installed on the instrument. The full spectrum capture enables the use of the novel unmixing algorithms for the further data analysis. Till now up to 45 different fluorescent labels could be resolved at same time, even when multiple markers are co-expressed on the same cell.
There are also three benchtop alignment-free multipurpose conventional analyzers. The laser configuration on BD LSR-Fortessa instrument allows for analysis of cell samples stained with 355, 405, 488, 561 and 640 nm excited dyes. A number of applications, including the multicolor analysis of cell phenotype, gene expression, cell cycle, and others may be performed. Agilent NovoCyte Penteon is equipped with five lasers (355, 405, 488, 561 and 640 nm) and 30 ultimate photodetectors, which provides an expanded set of capabilities that accommodate high-end and increasingly sophisticated multi-color flow cytometry assays with its laboratory automation platforms for FACS tubes (40-tube rack), cluster tubes and 24-, 48-, 96-, and 384-well plates. ThermoFisher Attune NxT is a conventional analyzer equipped with four lasers (405, 488, 561 and 637 nm) and 14 fluorescence channels. Using the unique acoustic focusing feature, the Autosampler enables Attune to run samples tubes or in plate formats, including 96-well, 384-well, and deep-wells with the high volume intake.
After orientation trainings (Virtual Classroom, “Beyond the Basics” Class, Pre-Hands-On-Consultation and Instrument Hands-On Training), Cytek Aurora, BD LSR-Fortessa, Agilent NovoCyte Penteon and ThermoFisher Attune NxT analyzers are available for use directly by the investigators.
By the collaboration with the Columbia University Stem Cell Initiative (CSCI) Flow Cytometry Core (FCC), FCRC facilitates access to ImageStream service for RU researchers to use the unique Cytek / AmnisⓇ ImageStreamX Mk II Imaging Cytometer. To access to the instrument, RU researchers need to request on PPMS system for initial internal consultation from FCRC Staff, followed by meeting with the CSCI FCC Director of Operations , Michael Kissner for the Experimental Design and Data Analysis Consultation to discuss experiment-specific details and schedule experiment(s).
Click to Enlarge.