How the antioxidant glutathione keeps mitochondria healthy
Kateryna Kon/Science Photo Library
If a delivery person leaves a package on your front step without pinging you, you likely won’t know it’s there. A hungry cell awaiting refuel is in a similar position. It has to be alerted to the presence of nutrients outside of the cell wall by a sensing mechanism so that a transporter protein can bring the nourishment inside.
The handful of these nutrient-sensing mechanisms thus far identified have had a profound impact on human health. One prime example is the discovery of the nutrient sensing mechanism for cholesterol, which led to the development of life-saving statin drugs (and the Nobel Prize).
These discoveries have focused on how an entire cell detects nutrients. But within every human cell are self-contained, membrane-bound organelles, all of which are equally in need of fuel to carry out important functions. Might they, then, have nutrient sensors of their own?
As described in a new paper published in Science, Kıvanç Birsoy and his colleagues in Rockefeller’s Laboratory of Metabolic Regulation and Genetics have discovered the first such sensor for an organelle—specifically mitochondria, the cell’s power center. The sensor is part of a protein that does triple duty: it senses, regulates, and delivers the antioxidant glutathione into the mitochondrial interior, where it plays critical roles in tamping down oxidizing reactions and maintaining appropriate iron levels.
“I believe this is going to be a very fruitful find,” says Birsoy. “Every time people have studied nutrient sensing, we’ve learned a lot about biology, and many drugs have been developed as a result.”
Antioxidant power
Glutathione is an antioxidant produced throughout the body that plays many important roles, including neutralizing unstable oxygen molecules called free radicals, which cause damage to DNA and cells if left unchecked. It also helps repair cellular damage and regulates cell proliferation, and its loss is associated with aging, neurodegeneration, and cancer. As a result, glutathione supplements have become increasingly popular as an over-the-counter approach to wellness.
The antioxidant is especially abundant in mitochondria, which cannot function without it. “As the respiratory organelle, mitochondria produces energy,” Birsoy notes. “But mitochondria can also the source of a lot of oxidative stress,” which has been implicated in cancer, diabetes, metabolic disorders, and heart and lung diseases, among others. If glutathione levels aren’t precisely maintained in mitochondria, all systems fail. None of us can survive without it.
But how glutathione actually enters mitochondria was unknown until 2021, when Birsoy and his team discovered that a transporter protein called SLC25A39 delivers the package. It also appeared to regulate the amount of glutathione. “When the antioxidants are low, the level of SLC25A39 increases, and when the antioxidant levels are high, the transport level goes down,” Birsoy says.
The findings strongly suggested that the mitochondria had some sort of way to detect and adjust these fluctuating levels. “Somehow mitochondria figures out how much antioxidant it has, and depending on that amount, it regulates the amount of antioxidant it lets inside,” he says.
Independent domains
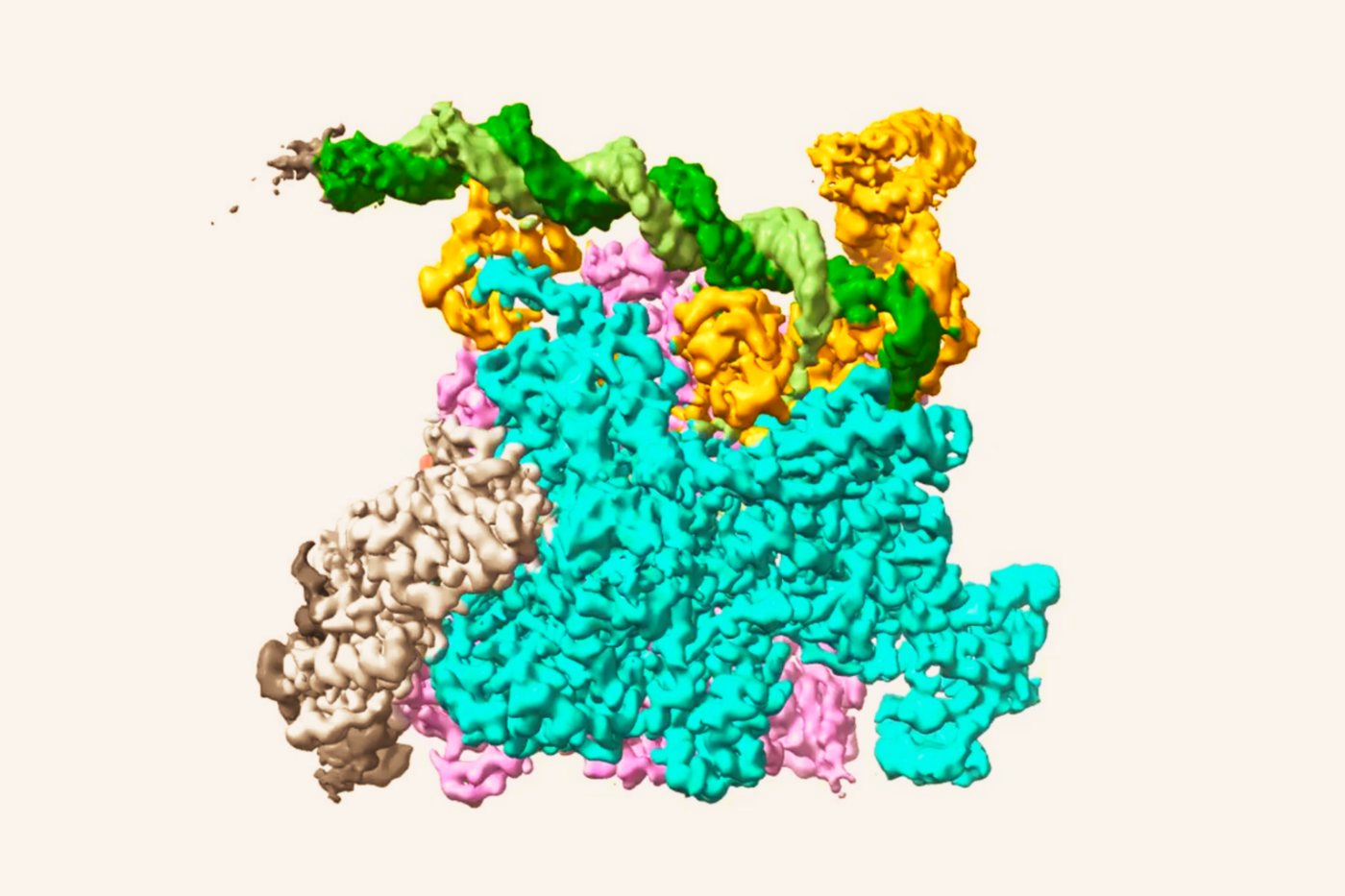
To ferret out how the mitochondria does it, the researchers used a combination of biochemical studies, computational methods, and genetic screens to discover that “SLC25A39 is both a sensor and a transporter at the same time,” Birsoy explains. “It has two completely independent domains. One domain senses the glutathione, and the other transports it.”
The protein’s unique structure may explain its abilities, says Birsoy. When Yuyang Liu, a graduate student in his lab and first author of the study, compared SLC25A39’s structure against others in the SLC family of transporters in the AlphaFold protein structure database, Liu spotted a unique extra loop in the protein. When they snipped it from the protein, its transporter abilities remained intact, but it lost the ability to sense glutathione. “Finding that interesting loop later led to our understanding of the sensing mechanism,” Birsoy says.
Iron worker
The study also bolsters the theory that glutathione is a “chaperone” for iron, which is required for virtually all functions within a cell, Birsoy says.
“Iron is not only the most abundant metal on Earth, it’s also the most abundant metal in our cells,” he says. But iron is also highly oxidative; without glutathione to keep it in line, it initiates oxidative stress in cells, causing damage. “We believe maintaining the glutathione-to-iron ratio is very important, because if you have too little glutathione, then iron becomes very reactive, and if you have too much glutathione, the iron will not be usable.” Their experiments determined that SLC25A39 carries a unique iron signature on its surface as part of the glutathione sensing mechanism.
Now that the researchers know how SLC25A39’s package delivery system operates, they can experiment with manipulating it. “This particular transporter protein is upregulated in a group of cancers,” Birsoy says. “People have tried to change overall glutathione levels, but now we have a way to change it in mitochondria without impacting other parts of the cell. This kind of targeted therapy could potentially lower the number of side effects that can come with altering glutathione levels across the whole body. I could see a lot of translational outcomes leveraging this new understanding.”