Gum disease may lie at the root of some arthritis flare-ups
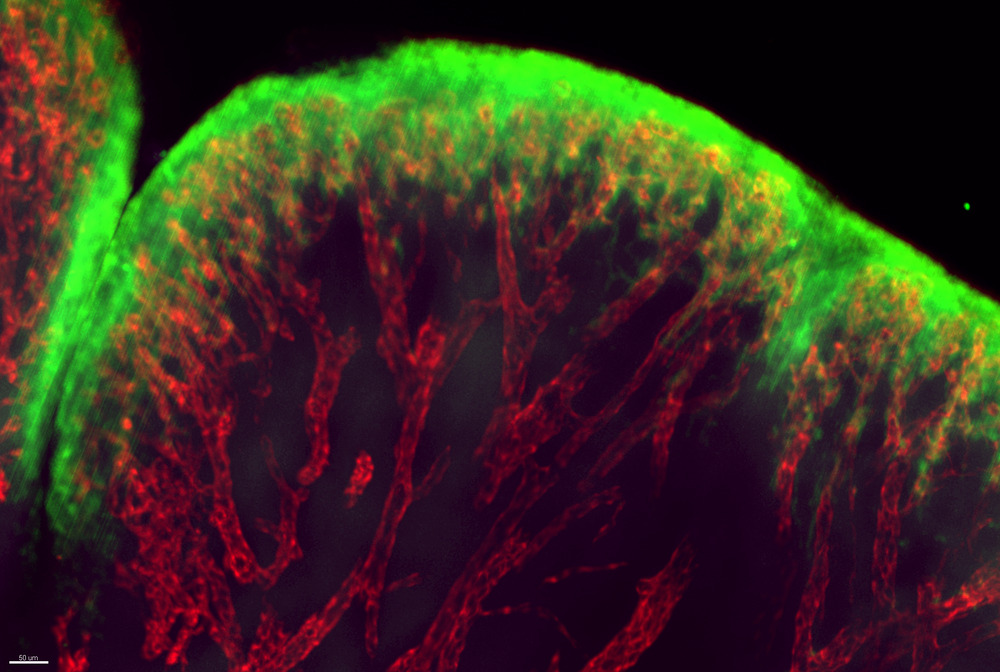
Blood vessels in the soft tissue between joints, during a rheumatoid arthritis flare-up.
It’s a well-documented medical mystery: Patients with gum disease are less likely to respond to rheumatoid arthritis treatments. But new research may help explain this link between gum disease and an otherwise disparate condition. The findings, published in Science Translational Medicine, suggests that breaches in damaged gums allow bacteria in the mouth to seep into the bloodstream, activating an immune response that ultimately pivots to target the body’s own proteins and causes arthritis flare-ups.
“If oral bacteria are getting in and repeatedly triggering immune responses relevant to rheumatoid arthritis, that could make it harder to treat,” says Dana Orange, associate professor of clinical investigation in the laboratory of Robert B. Darnell at The Rockefeller University. “When doctors encounter arthritis patients who do not respond to treatment, it would be worth it to make sure they aren’t missing an underlying gum disease, which is quite treatable.”
The Darnell lab had been following a small group of arthritis patients over the course of several years, collecting weekly blood samples and looking for changes in gene expression to help explain why painful flare-ups occur, when they noticed a surprising trend. Two of their patients, who had moderate to severe periodontal disease, had repeated episodes of oral bacteria in their bloodstreams, even when they weren’t having dental work.
Orange knew that rheumatoid arthritis patients generally have autoantibodies in their bloodstream (rheumatoid arthritis is an auto-immune disease, wherein antibodies attack the body’s own proteins and peptides). In many cases, autoantibodies take specific aim at proteins bearing the signs of citrullination, a process by which one amino acid in the protein is converted into a different one.
Upon further examination, the team discovered that the oral bacteria they detected in the blood are also citrullinated in the mouth, much like the proteins targeted by autoantibodies in arthritis. They then demonstrated that the same autoantibodies that take potshots at the body’s citrullinated proteins activate in response to citrullinated bacteria.
The results may explain why arthritis treatments do not work as well in patients with gum disease. If the gums are continuously releasing immune triggers into the bloodstream, treating arthritis without first solving the periodontal problem is like trying to haul water out of a ship without first plugging up its leaks.
“Gum disease is quite curable; rheumatoid arthritis can be much more difficult to treat,” Orange says. “Our results indicate that periodontal disease leads to leaky gums that allow oral bacteria to enter the blood repeatedly. This level of oral bacteria in blood doesn’t cause obvious symptoms, so the patients were not aware this was happening, but they do trigger inflammatory and auto-antibody responses that are highly relevant to rheumatoid arthritis.”
These findings also demonstrate the importance of conducting long-term research to better understand chronic diseases. The present study would not have been possible without a unique initiative, pioneered by Orange and Darnell several years ago, that empowers arthritis patients to collect their own blood samples at home with a finger-prick kit and mail weekly samples to Rockefeller. The lab now has several years of data to help track what happens in the blood right before an arthritis flare.
“Without having weekly blood samples for at least a year, we wouldn’t have been able to find out what was happening before the patients had symptoms of their flares,” Orange says. “Our study revealed a plausible mechanism to explain why rheumatoid arthritis patients with periodontal disease do not respond well to treatment—something very hard to capture without long-term monitoring.”