New approach exposes 3D structure of Alzheimer’s proteins within the brain
Alzheimer’s disease clouds memory, dims the mind, and distorts behavior. Its ravages also show up within the physical structure of the brain, perhaps most prominently as sticky clumps of a naturally occurring but harmful protein called amyloid-β.
A team at The Rockefeller University used a new approach, known as iDISCO, that makes brain tissue transparent to permit the capture of detailed three-dimensional views of amyloid-β plaques within mouse and human brains. Their results are described July 14 in Cell Reports.
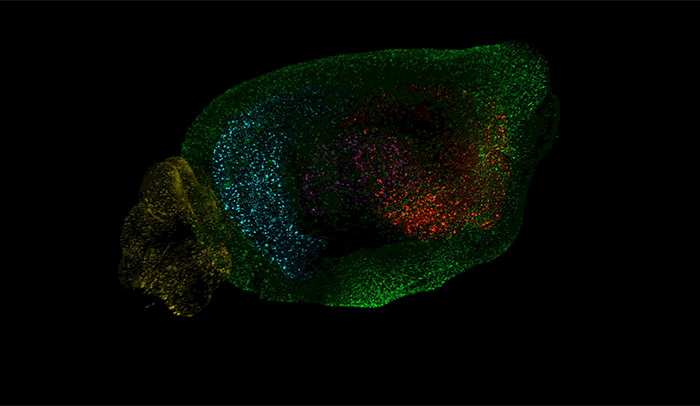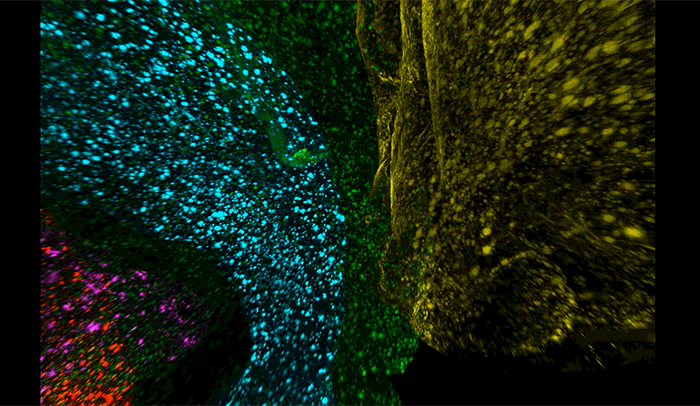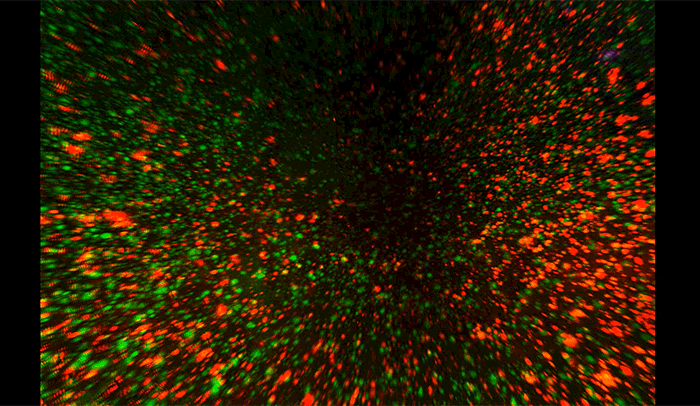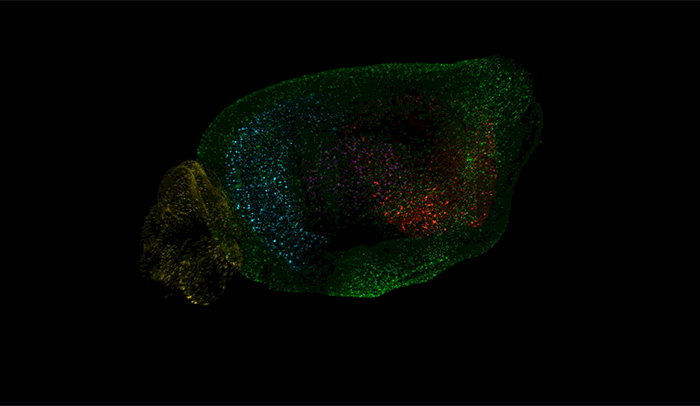
Above, amyloid-beta plaques appear as speckles within half of a brain of a mouse used to study the disease. Brain regions are coded by color, with the cerebellum in yellow, hippocampus in blue, thalamus in purple, striatum in red, and cortex in green.
Thomas Liebmann, the lead author, and his colleagues also used iDISCO to examine small blocks of frozen tissue from deceased Alzheimer’s patients, and found that the human plaques were larger and more complex than those from the mice. This discovery could aid researchers in establishing different categories for the disease based on a patient’s symptoms and the plaques within his or her brain. The relationship between plaques and dementia is poorly understood currently; the two do not always occur together.
“A better understanding of these plaques, as well as other key features of Alzheimer’s in the brain, might contribute to efforts to develop better targeted drugs, or allow us to rethink the drugs we have now—that’s what we hope for,” says corresponding author Marc Flajolet, a research assistant professor in Paul Greengard’s Laboratory of Molecular and Cellular Neuroscience.
This work was done in collaboration with Marc Tessier-Lavigne’s Laboratory of Brain Development and Repair and with assistance from the university’s Bio-Imaging Resource Center.
Funding statement: This work was supported by the Fisher Center for Alzheimer’s Research Foundation, the Cure Alzheimer’s Fund, the NIH (NIA grant AG09464), and the Empire State Stem Cell Fund (NYSDOH contract #C023046).
 Cell Reports online: July 14, 2016 Cell Reports online: July 14, 2016Three-Dimensional Study of Alzheimer’s Disease Hallmarks Using the iDISCO Clearing Method Thomas Liebmann, Nicolas Renier, Karima Bettayeb, Paul Greengard, Marc Tessier-Lavigne, and Marc Flajolet |